Abstract
The inclusion of infants in the SARS-CoV-2 vaccine roll-out is important to prevent severe complications of pediatric SARS-CoV-2 infections and to limit transmission and could possibly be implemented via the global pediatric vaccine schedule. However, age-dependent differences in immune function require careful evaluation of novel vaccines in the pediatric population. Toward this goal, we assessed the safety and immunogenicity of two SARS-CoV-2 vaccines. Two groups of 8 infant rhesus macaques (RMs) were immunized intramuscularly at weeks 0 and 4 with stabilized prefusion SARS-CoV-2 S-2P spike (S) protein encoded by mRNA encapsulated in lipid nanoparticles (mRNA-LNP) or the purified S protein mixed with 3M-052, a synthetic TLR7/8 agonist in a squalene emulsion (Protein+3M-052-SE). Neither vaccine induced adverse effects. Both vaccines elicited high magnitude IgG binding to RBD, N terminus domain, S1, and S2, ACE2 blocking activity, and high neutralizing antibody titers, all peaking at week 6. S-specific memory B cells were detected by week 4 and S-specific T cell responses were dominated by the production of IL-17, IFN-γ, or TNF-α. Antibody and cellular responses were stable through week 22. The immune responses for the mRNA-LNP vaccine were of a similar magnitude to those elicited by the Moderna mRNA-1273 vaccine in adults. The S-2P mRNA-LNP and Protein-3M-052-SE vaccines were well-tolerated and highly immunogenic in infant RMs, providing proof-of concept for a pediatric SARS-CoV-2 vaccine with the potential for durable immunity that might decrease the transmission of SARS-CoV-2 and mitigate the ongoing health and socioeconomic impacts of COVID-19.
INTRODUCTION
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has infected hundreds of millions of people worldwide and caused over 3.5 million deaths since its emergence in 2019. The need for safe and effective measures to limit transmission and mitigate public health and socioeconomic impacts of SARS-CoV-2 infection has prompted unprecedented vaccine development of promising candidates. In fact two messenger RNA (mRNA) vaccines, mRNA-1273 (Moderna) (1, 2) and BNT162b2 (Pfizer-BioNTech) (3, 4), are authorized for emergency use in the United States to prevent SARS-CoV-2 infection in adults. Both are safe, and induce neutralizing antibodies and up to 95% protection from disease. Additionally, the Oxford/AstraZeneca adenovirus-based ChAdOx1 nCoV-19 (AZD1222) (5), the Johnson & Johnson Ad26.COV2.S vaccine (JNJ-78436735) (6), and the first protein-based SARS-CoV-2 vaccine, Novavax NVX-CoV2373, adjuvanted with saponin-based Matrix-M (7, 8), are approved for use in human adults.
Ethical and safety risks warrant a careful evaluation of novel vaccines in the pediatric population. Generally, vaccine testing is performed in an age de-escalation manner, starting with adults, followed by adolescents, with children and infants being last (9). The BNT162b2 mRNA vaccine is now approved for use in adolescents 12 years and older, with trials for children age 6 to 12 already underway (NCT04816643). The Moderna mRNA-1273 vaccine is close to approval for use in children 12 years and older. Importantly, Moderna and Pfizer both have initiated clinical trials that will include infant as young as 6 months (NCT04796896 and NCT04816643, respectively). The important epidemiologic impact of pediatric SARS-CoV-2 vaccination lies in limiting transmission and ease of implementation via the global pediatric vaccine schedule.
Early in the pandemic, vaccines to prevent SARS-CoV-2 infection in children were not a priority because of apparent low infection and disease rates. In most children, SARS-CoV-2 infection causes only relatively mild disease. Nonetheless, some children develop severe symptoms, such as the multisystem inflammatory syndrome, requiring hospitalization and sometimes leading to death (10–14). Importantly, severe complications of SARS-CoV-2 infection disproportionally affect children of ethnic and racial minorities, amplifying health disparities in pediatric care in the United States (15, 16). While children may transmit less efficiently than adults, virus transmission by children, even when asymptomatic, is documented (17, 18). Therefore, children have the propensity to become a major viral reservoir if pediatric vaccination stalls. The latter potential is especially concerning because new SARS-CoV-2 variants with increased transmission rates are emerging and SARS-CoV-2 has become more likely to persist on the population level. Although data are scarce, increased pediatric infection rates that appeared to coincide with the emergence of new variants, have been reported (19, 20). There is ample precedence for the beneficial impact of pediatric vaccination.
Children have suffered from the SARS-CoV-2 pandemic in many additional ways. We will not know the exact burden of the loss of education due to closure of schools and virtual learning until years from now. School closure meant that many children were deprived of social interactions, a food source, and a safe place (21–23). There has been an epidemic rise in depression and anxiety in children, and the long-term mental health burden of these diverse factors is difficult to assess. School closure also translated to loss of work for many parents, especially women. A pediatric SARS-CoV-2 vaccine could mitigate at least some of these societal costs.
These combined data emphasize the importance of getting children vaccinated for SARS-CoV-2 (24–27). However, vaccine-elicited immunity differs between adults and infants (reviewed in (28)), with impaired responses to carbohydrate antigen vaccines (29), but higher magnitude humoral immunity to subunit-based vaccines (e.g., Hepatitis B) (30) early in life. Thus, the evaluation of SARS-CoV-2 vaccine immunogenicity in infants is critical. Toward this goal, the present study was designed to evaluate the safety and immunogenicity of stabilized prefusion SARS-CoV-2 Spike vaccines delivered as lipid nanoparticle-encapsulated mRNA or an adjuvanted subunit protein in a relevant animal model. Non-human primates (NHP), including rhesus macaques (RMs), are an important model for SARS-CoV-2 studies given the similarities to humans in pathogenesis and host immune responses (31). Indeed, results from adult NHP studies of human SARS-CoV-2 vaccine candidates (32–35) strongly correlate with clinical trial outcomes, and infant NHP models of other infectious diseases closely mirror responses in human infants (36, 37). Data by our group support that infant vaccine-elicited antibody against HIV envelope can be of higher magnitude than those elicited by adults, (38) We also demonstrated that infant RMs generate high magnitude antibody responses to HIV Env protein vaccination, (39) and that peripheral and mucosal B cell responses can be boosted later in life. (40)
The primary goal of the current study was to inform human pediatric trials and aid in fast-tracking pediatric SARS-CoV-2 vaccines for inclusion in the early life vaccine schedule by providing proof-of-concept that the two SARS-CoV-2 vaccines are safe and immunogenic in infant RMs. A secondary goal of our study was to assess the durability of vaccine-induced immune responses as data are lacking on this. Despite some caveats inherent to comparisons between studies with differences in study design, the immune responses in infant RMs in response to the two SARS-CoV-2 vaccines tested appear to be within the range of vaccine-induced immune responses reported for adult RMs (32, 41) and humans (2, 42, 43).
RESULTS
Study design and vaccine safety
Infant RMs at a median age of 2.2 months, corresponding to 9-month old human infants (44), were immunized at weeks 0 and 4 with 30 μg mRNA encoding stabilized prefusion SARS-CoV-2 S-2P protein in lipid nanoparticles (mRNA-LNP; n=8) or with 15 μg S-2P mixed with 3M-052-SE, a TLR7/8 agonist in a stable emulsion (Protein+3M-052-SE; n=8) (Fig. 1, Table 1). Blood and saliva were collected before vaccination (week 0), 4 weeks after the first dose of the vaccine (week 4), 2 weeks after the vaccine boost (week 6), and at weeks 8, 14, 18 and 22. Lymph nodes were sampled at week 6 (Fig. 1).
Study Design: evaluation of immunogenicity of two SARS-CoV-2 vaccines in infant rhesus macaques. Infant rhesus macaques (median age of 2.2 months at study initiation) were immunized at 0 and 4 weeks with either 30 μg mRNA encoding S-2P (Vaccine Research Center, NIH) in lipid nanoparticles (mRNA-LNP) or 15 μg S-2P protein formulated with 3M-052 adjuvant, a TLR7/8 agonist, as a stable emulsion (3M-052-SE). Each group consisted of 8 animals. Blood and saliva samples were collected at weeks 0, 4, 6, 8, 14, 18 and 22, and lymph node biopsies were obtained at week 6.
” data-icon-position=”” data-hide-link-title=”0″>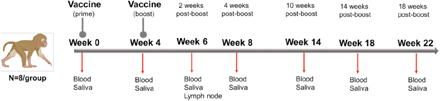

Study Design: evaluation of immunogenicity of two SARS-CoV-2 vaccines in infant rhesus macaques. Infant rhesus macaques (median age of 2.2 months at study initiation) were immunized at 0 and 4 weeks with either 30 μg mRNA encoding S-2P (Vaccine Research Center, NIH) in lipid nanoparticles (mRNA-LNP) or 15 μg S-2P protein formulated with 3M-052 adjuvant, a TLR7/8 agonist, as a stable emulsion (3M-052-SE). Each group consisted of 8 animals. Blood and saliva samples were collected at weeks 0, 4, 6, 8, 14, 18 and 22, and lymph node biopsies were obtained at week 6.
Animals were monitored daily for adverse events. No local injection site or systemic adverse reactions were observed. Animals had normal blood metrics (Supplementary Table S1). Importantly, the animals gained weight consistent with adequate fluid and nutrition uptake and with normal infant growth (Supplementary Fig. S1) (45). A major safety concern in the implementation of a pediatric SARS-CoV-2 vaccine stems from adverse events observed with an earlier Respiratory Syncytial Virus vaccine, that were partially attributed to T helper 2 (TH2)-biased responses (46, 47). We therefore tested plasma samples prior to or following immunization for changes in TH2 (IL-4, IL-13) or TH1 (IL-2, IFN-γ) cytokines (Fig. S2). Animal RM3 of the protein group (Table 1) tested positive for all cytokines at both time points; cytokines were also detected in the mRNA-LNP recipient RM11 at week 0, but fell below the limit of quantification by week 6. In general, neither vaccine appeared to induce systemic TH2 or TH1 responses (Fig. S2), further supporting a good safety profile.
Infant plasma and salivary antibody responses to SARS-CoV-2 vaccination
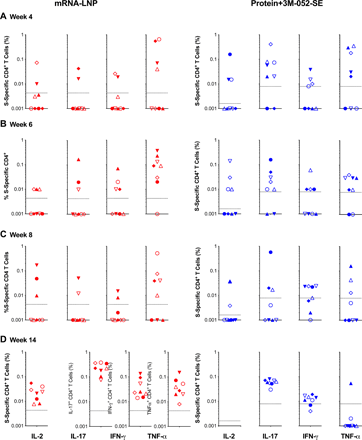
Plasma IgG binding to the S-2P protein was observed after the vaccine prime for both mRNA-LNP and Protein+3M-052-SE vaccines and increased after the second immunization (Fig. 2). Although antibody levels dropped by week 8, there was only a slight decline of S-2P-specific IgG from week 8 to week 22 for mRNA-LNP (median AUC and 95% confidence intervals: 7.0 [6.2, 8.3] to 5.4 [4.2, 7.7]) or Protein+3M-052-SE (11.0 [9.5, 11.7] to 9.4 [8.6, 10.5] vaccinees (Fig. 2A). Longitudinal S-specific plasma IgG data reported as EC50 or endpoint dilution titers followed a similar trend (Fig. S3). As the D614G virus variant had become dominant in the USA at the study initiation, we confirmed that IgG binding to S-2P D614G was similar to that of D614 at weeks 6 and 14 (Fig. S4). Plasma IgG antibodies were directed against multiple spike domains and persisted throughout the study, with robust binding to Spike regions 1 and 2 (S1, S2), Receptor binding domain (RBD), and N-terminal domain (NTD) (Fig. 2B). RBD-specific salivary IgG from mRNA-LNP recipients peaked at a median of 16.6 ng RBD-specific IgG per μg of total IgG at week 6 (Fig. 2C). In the Protein+3M-052-SE group, median salivary RBD-specific IgG peaked at 98.2 ng/μg IgG after the second vaccination and remained detectable throughout the study (Fig. 2C). Saliva RBD-specific IgA responses were much lower (Fig. S5). Spike-specific IgM and IgA in plasma were low or undetectable (Fig. S5). In the mRNA-LNP group, IgM increased through week 14, but the absorbance readings for IgM were considered low, measured at a 1:10 sample dilution (Fig. S5).
Plasma and saliva were collected before vaccination (week 0), at week 4 – prior to the 2nd dose-, two weeks post 2nd dose (week 6), and at weeks 8, 14, 18 and 22 from infant RM vaccinated with 30 μg mRNA encoding S-2P spike protein in lipid nanoparticles (n=8; red) or with 15 μg prefusion SARS-CoV-2 S-2P spike protein formulated with 3M-052 adjuvant (n=8; blue). (A): S-2P protein-specific antibody responses were measured by enzyme-linked immunosorbent assay (ELISA). Serial dilutions of plasma starting at 1:40 were assayed for IgG binding to SARS-CoV-2 spike. Data are reported as log10 area under the curve (AUC) values. (B): Antibody epitope specificity measured by BAMA. Plasma was diluted 1:10,000 to measure binding to different domains of the spike protein, including the full-length S protein, S1, RBD, NTD, and S2. Binding antibody responses are reported as log10 transformed mean fluorescence intensity (MFI) after subtraction of background values. (C): Salivary RBD-specific IgG was measured by binding antigen multiplex assay (BAMA) using serial dilutions of saliva and responses are reported as RBD-specific IgG (ng) / total IgG (μg). Different symbols represent individual animals (Table 1). Arrows in Panel A indicate times of immunizations.
” data-icon-position=”” data-hide-link-title=”0″>

Plasma and saliva were collected before vaccination (week 0), at week 4 – prior to the 2nd dose-, two weeks post 2nd dose (week 6), and at weeks 8, 14, 18 and 22 from infant RM vaccinated with 30 μg mRNA encoding S-2P spike protein in lipid nanoparticles (n=8; red) or with 15 μg prefusion SARS-CoV-2 S-2P spike protein formulated with 3M-052 adjuvant (n=8; blue). (A): S-2P protein-specific antibody responses were measured by enzyme-linked immunosorbent assay (ELISA). Serial dilutions of plasma starting at 1:40 were assayed for IgG binding to SARS-CoV-2 spike. Data are reported as log10 area under the curve (AUC) values. (B): Antibody epitope specificity measured by BAMA. Plasma was diluted 1:10,000 to measure binding to different domains of the spike protein, including the full-length S protein, S1, RBD, NTD, and S2. Binding antibody responses are reported as log10 transformed mean fluorescence intensity (MFI) after subtraction of background values. (C): Salivary RBD-specific IgG was measured by binding antigen multiplex assay (BAMA) using serial dilutions of saliva and responses are reported as RBD-specific IgG (ng) / total IgG (μg). Different symbols represent individual animals (Table 1). Arrows in Panel A indicate times of immunizations.
Function of vaccine-induced plasma antibodies
We next evaluated the capacity of plasma antibodies to block entry of SARS-CoV-2 into human cells using a RBD-ACE2 blocking assay at 1:10 and 1:40 plasma dilutions. Antibodies elicited by the mRNA-LNP vaccine completely blocked RBD-ACE2 interaction at week 6 at 1:10 and 1:40 dilutions. Greater than 80% blocking was achieved at 1:10 dilution until week 14, dropping for some animals after week 18 (Fig. 3A). The Protein+3M-052-SE vaccine induced antibodies that mediated 100% RBD-ACE2 blocking after the second immunization (Fig. 3A). At a 1:40 plasma dilution, the RBD-ACE2 blocking function decreased over time, but was still detectable at week 22 (Fig. S6).
SARS-CoV-2 vaccine-elicited functional antibody responses in infant rhesus macaques. (A): The capacity of plasma antibodies to mediate blocking of the RBD-ACE2 interaction was measured with an ELISA-based ACE2 blocking assay at 1:10 plasma dilution. Data are reported as %ACE2 blocking. (B-C): Neutralization capacity was measured using Spike D614G-pseudotyped viruses in 293T/ACE2 cells (B) and whole virus (D614G) assay with Vero E6 cells (C); results are expressed as reciprocal 80% inhibitory dilution (ID80). Grey dotted lines represent detection cut-off. Different symbols represent individual animals (Table 1). Longitudinal data for each animal in the mRNA-LNP (red) or Protein+3M-052-SE group (blue) are represented by separate lines. Arrows in Panel C indicate times of immunizations.
” data-icon-position=”” data-hide-link-title=”0″>

SARS-CoV-2 vaccine-elicited functional antibody responses in infant rhesus macaques. (A): The capacity of plasma antibodies to mediate blocking of the RBD-ACE2 interaction was measured with an ELISA-based ACE2 blocking assay at 1:10 plasma dilution. Data are reported as %ACE2 blocking. (B-C): Neutralization capacity was measured using Spike D614G-pseudotyped viruses in 293T/ACE2 cells (B) and whole virus (D614G) assay with Vero E6 cells (C); results are expressed as reciprocal 80% inhibitory dilution (ID80). Grey dotted lines represent detection cut-off. Different symbols represent individual animals (Table 1). Longitudinal data for each animal in the mRNA-LNP (red) or Protein+3M-052-SE group (blue) are represented by separate lines. Arrows in Panel C indicate times of immunizations.
Neutralizing antibodies, measured by a pseudovirus assay, were detected in all mRNA-LNP vaccinated animals and in 7 of 8 animals of the Protein+3M-052-SE group after the first vaccination, and further increased after the boost. Peak median infectious dose 80 (ID80) titers were observed at week 6 and reached 1,179 in mRNA-LNP and 13,687 in Protein+3M-052-SE animals (Fig. 3B). Similar to S-specific plasma IgG levels, and consistent with antibody kinetics after vaccination, median ID80 neutralization titers decreased following the peak response. Neutralization titers appeared to stabilize around week 18, and median ID80 titers at week 22 remained 2.6-fold or 14.2-fold higher in the mRNA-LNP or Protein+3M-052-SE group, respectively, than after the first vaccination (week 4) (Fig. 3B). Although definite immune correlates of protection after vaccination still need to be conclusively determined, we modeled a bi-phasic antibody decline for both vaccines to reach a putative protective neutralizing Ab titer, estimated at a reciprocal dilution of 100 (41, 48). Applying this assumption, mRNA-LNP vaccinees maintained a protective neutralizing Ab titer for approximately 46 weeks and protein vaccine recipients for approximately 74 weeks (Fig. S7).
Neutralizing antibody kinetics in the whole virus neutralization assay followed a similar trend (Fig. 3C). The animal (RM 1) of the Protein+3M-052-SE group that did not have detectable neutralizing antibodies by week 4 in the pseudovirus neutralization assay, also only had a slight increase in the ID80 neutralizing antibody titer using the live virus assay (week 0: 129; week 4: 223). Overall, ID50 and ID80 titers of both assays strongly correlated with each other (r = 0.644, p=0.008 or r=0.785, p=0.0005, respectively Fig. S8). However, infant RMs already had low titer neutralizing antibodies at day 0 when we applied the whole virion neutralization assay (Fig. 3C). As infants could have passively acquired antibodies from their mothers, we measured neutralizing antibodies in the sera of their unvaccinated dams. Although detectable, neutralizing antibody titers in dams did not correlate to infant neutralization titers before (ID80: r=0.011, p=0.96) or after vaccination (ID80: r=-0.056, p=0.83) (Fig. S9), most likely because infant rhesus macaques were already 2 months old at study initiation and maternal antibodies had waned.
Vaccine-elicited B cell responses
Consistent with the induction of plasma S-specific IgG, S-specific memory CD27+ B cell were detectable in the blood of both vaccine groups at week 4 and peaked at week 6 (median: 0.75% mRNA-LNP; 3.12%, Protein+3M-052-SE). At week 14, median memory B cell frequencies were lower (0.19% and 0.13% in mRNA-LNP or Protein+3M-052-SE vaccinees, respectively), but did not differ from those at week 4 (Fig. 4A). In lymph nodes (LNs), we measured total germinal center (GC) B cells and memory B cells and then determined the number of S-specific memory B cells (Fig. 4B). In both vaccine groups robust memory B cell populations (median 3.78% mRNA-LNP and 1.96% Protein-3M-052) were present (Fig. 4B). Induction of S-specific B cells was confirmed by assessing S-specific antibody secreting cells (ASC). In the blood, peak responses were observed at week 6 in the mRNA-LNP group (median: 154 ASC/million cells) and at week 8 in the Protein+3M-052-SE group (median: 85 ASC/106 cells) (Fig. 4C). In draining LNs, mRNA-LNP and Protein+3M-052-SE vaccinees had median 6 or 952 ASC/106 cells, respectively (Fig. 4D).
(A) CD20+CD27+ memory B cells that co-stained with fluorochrome-conjugated SARS-CoV-2 spike protein in mRNA-LNP (red) or Protein+3M-052-SE (blue) vaccinees in blood. Frequencies are expressed as percent of total memory B cells. The gating strategy is provided in Supplementary Figure S14. (B): In lymph nodes, we determined total Bcl-6+Ki-67+ Germinal Center (GC) B cells and CD27+ memory B cells as percent of total CD20+ B cells (left panel) (see Fig. S15 for gating strategy) and also the percent of S-specific memory B cells (right panel). (C) Antibody secreting cell (ASC) as measured by B cell ELISpot in PBMC from mRNA-LNP or Protein+3M-052-SE vaccinees, while (D) is showing mRNA-LNP and Protein+3M-052-SE ASC responses, respectively, in LN at week 6. Different symbols represent individual animals (Table 1). Solid lines represent median values.
” data-icon-position=”” data-hide-link-title=”0″>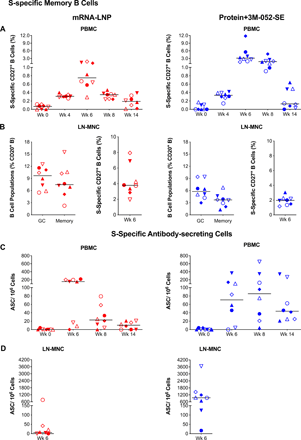

(A) CD20+CD27+ memory B cells that co-stained with fluorochrome-conjugated SARS-CoV-2 spike protein in mRNA-LNP (red) or Protein+3M-052-SE (blue) vaccinees in blood. Frequencies are expressed as percent of total memory B cells. The gating strategy is provided in Supplementary Figure S14. (B): In lymph nodes, we determined total Bcl-6+Ki-67+ Germinal Center (GC) B cells and CD27+ memory B cells as percent of total CD20+ B cells (left panel) (see Fig. S15 for gating strategy) and also the percent of S-specific memory B cells (right panel). (C) Antibody secreting cell (ASC) as measured by B cell ELISpot in PBMC from mRNA-LNP or Protein+3M-052-SE vaccinees, while (D) is showing mRNA-LNP and Protein+3M-052-SE ASC responses, respectively, in LN at week 6. Different symbols represent individual animals (Table 1). Solid lines represent median values.
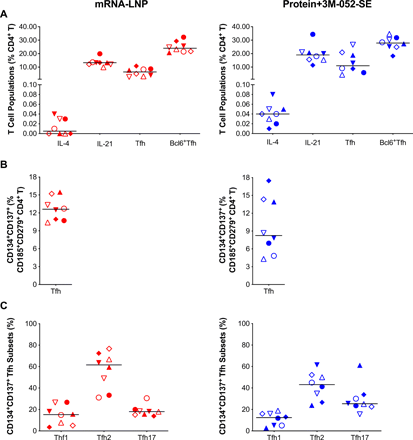
In addition, we analyzed LN samples for canonical T follicular helper (TFH) (CD185/CXCR5+CD279/PD-1+) cells and for IL-4 or IL-21 producing CD4+ T cells that support GC B cell differentiation (Fig. 5A). SEB-activated TFH (CD185+CD279+CD134+CD137+) (49) (Fig. 5B) were categorized into TFH1, TFH2 and TFH17 subsets based on expression of CXCR3/CD183 and/or CCR6/CD196 (50) (Fig. 5C). We found no correlations between bcl6+Ki67+ GC B cells or bcl6+TFH (Fig. S10A, B), potentially because we assessed these cell populations in an antigen non-specific manner. However, CD4+IL21+ cells correlated with pseudovirus neutralizing antibody ID50 at week 6 (r=0.749, p=0.001) (Fig. S10C).
Immunophenotype of lymph node T cell population two weeks post-boost. (A): CD4+ T-cells positive for IL-4 or IL-21, follicular T helper cell (Tfh) markers CD279/PD-1 and CD185/CXCR5, or Bcl6+ Tfh were measured and are represented as percent of total LN CD4+ T cells. The gating strategy for these panels is provided in Figure S15. (B): SEB activated Tfh frequencies assessed by the Activation-Induced Marker (AIM) Assay: CXCR5+CD185+ cells that co-expressed CD134 and CD137 for Protein+3M-052-SE and mRNA-LNP, respectively. (C) The frequency of CXCR5+CD185+CD134+CD137+CXCR3+CD196– Tfh1, CXCR3–CCR6– Tfh2 and CXCR3–CD196+ Tfh17 cells is shown. The gating strategy for these populations is given in Fig. S17. Red symbols: mRNA-LNP, blue symbols: Protein+3M-052-SE. Different symbols represent individual animals (Table 1). Solid lines define the median.
” data-icon-position=”” data-hide-link-title=”0″>